AliveCor BioPharma partners with pharmaceutical companies, research organizations, and clinical trials to remotely capture critical heart data, anytime and anywhere.
In partnership with Clinical Research Organizations (CROs) and pharmaceutical companies, AliveCor removes barriers to capturing study data by enabling easy remote cardiac monitoring for phase I-IV clinical trials.
Because certain medications may have potential adverse effects on patients, such as QT prolongation, many healthcare professionals reconsider prescribing them despite some medications having a higher efficacy than their alternatives. AliveCor’s Cardiac Safety Monitoring (CSM) programs are implemented to capture critical heart data prior to and during patient treatment to help physicians make informed medical decisions.
To help improve the delivery of life-changing medicines, AliveCor BioPharma enables ongoing patient monitoring of certain cardiac conditions. Our remote monitoring devices, apps, and services can be used for patient screening, disease monitoring, medication management and more.
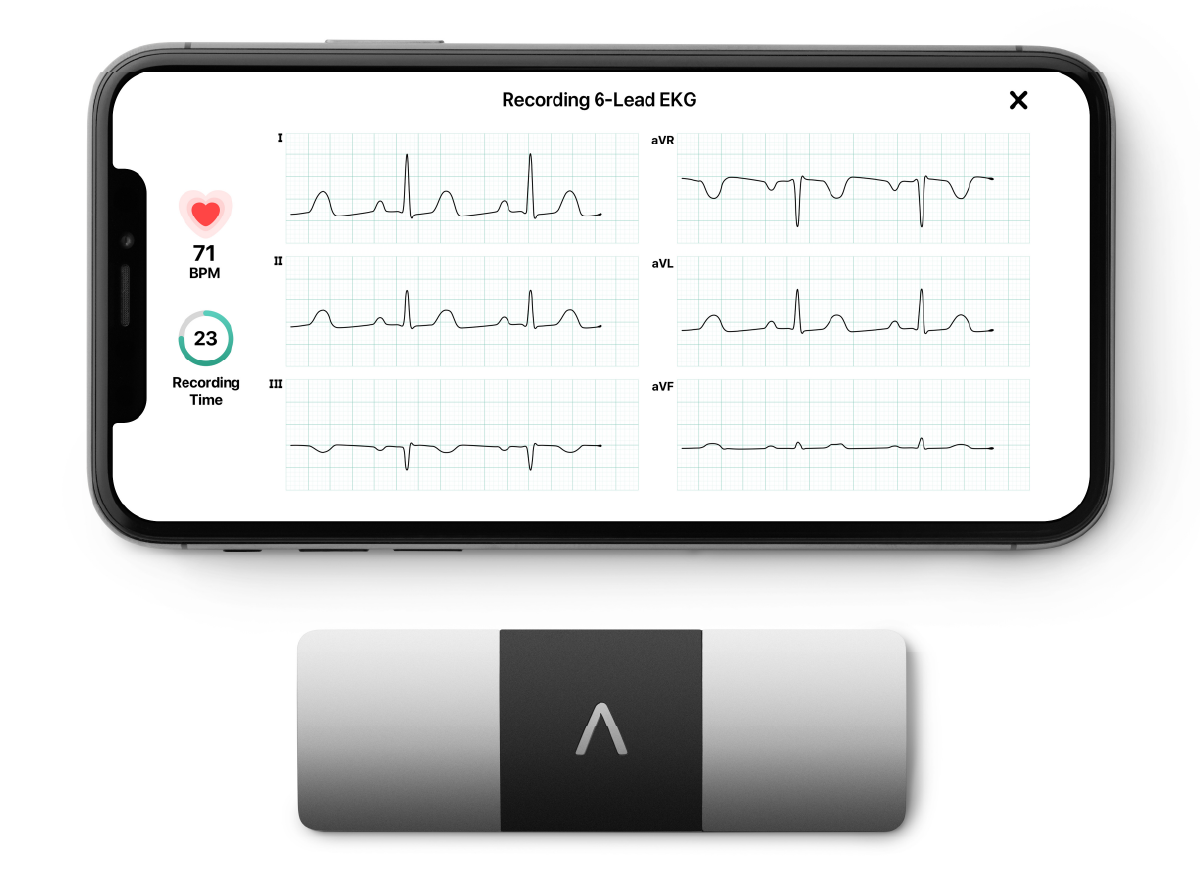
KardiaMobile 6L – The world’s first-and-only FDA-cleared, 6-lead personal ECG. With no wires, patches, or gels, KardiaMobile 6L captures six leads of medical-grade heart data onsite or remotely.

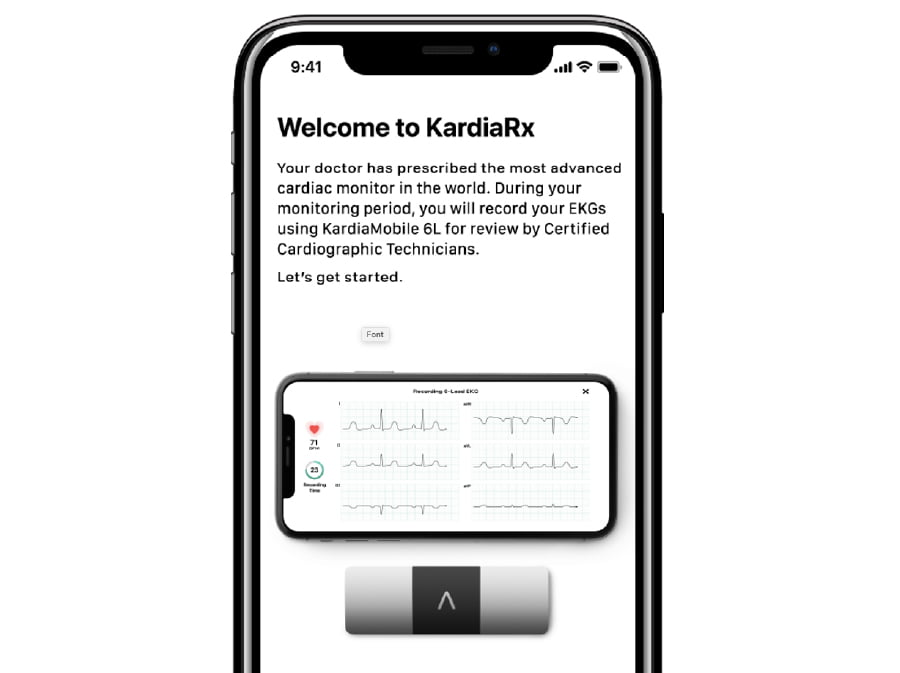
KardiaRx – Connect to your patients remotely. KardiaMobile 6L pairs to KardiaRx, a smartphone app that allows patients to record cardiac symptoms and capture 6-leads of heart data anytime, anywhere.
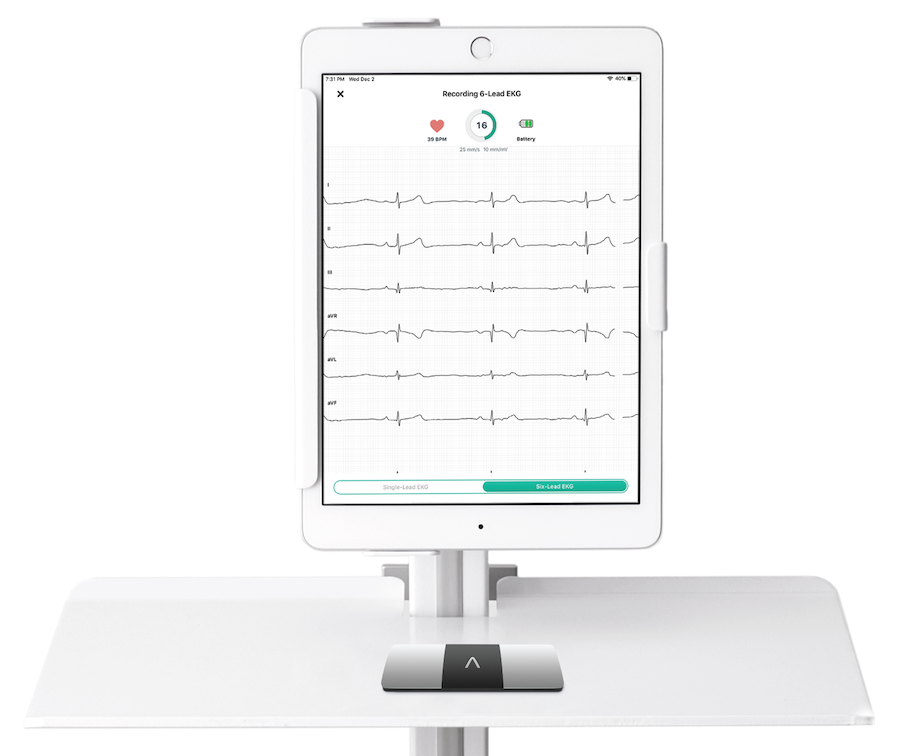
KardiaStation – Record medical-grade, 6-lead ECGs using KardiaStation, an app for medical-professionals to record ECGs onsite without the need for a 12-lead ECG machine. Receive instant FDA-cleared Kardia Determinations including possible atrial fibrillation, bradycardia, tachycardia, PVCs and more in just 30 seconds.
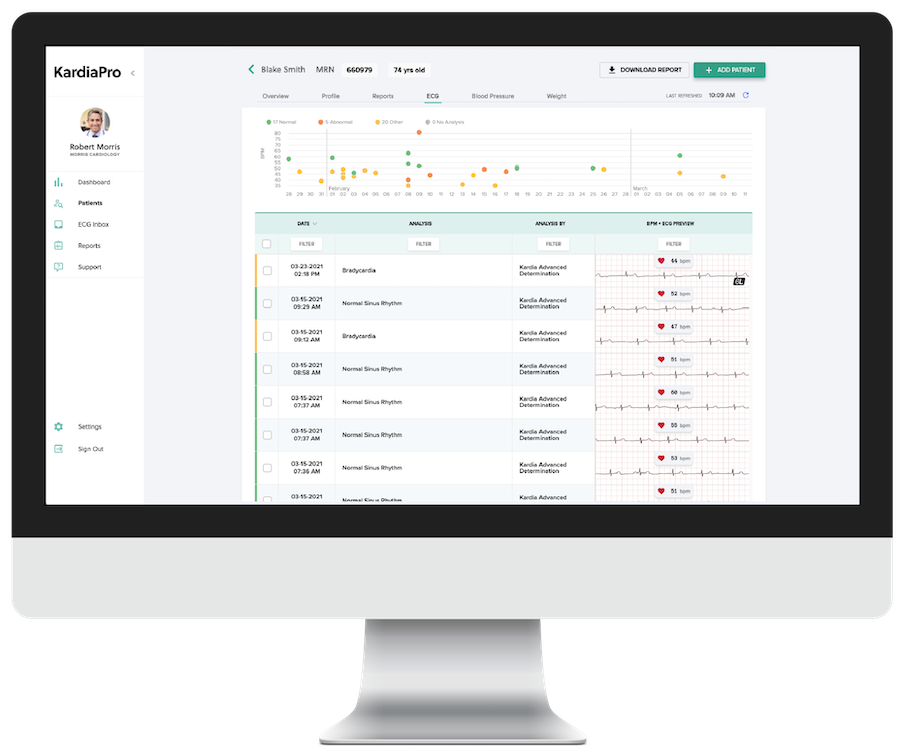
KardiaPro – Manage and review ECG data captured with KardiaMobile 6L on our HIPAA compliant, SOC-2 certified, web-based portal.
©2022 Copyright AliveCor in Hong Kong and Macau All Rights Reserved. The website is operated by Mellica Ltd., authorized distributior of AliverCor in HongKong and Macau.
AliveCor Labs